Education:
- PhD School of Biomedical Sciences, Kent State University, OH
- MA Biological Anthropology, Kent State University, OH
- BA Anthropology, University of South Florida
Professional Experience:
- Associate Scientist/Assistant Professor, Sanford Research Center/University of South Dakota
- Research Associate (Assistant Professor), The University of Chicago
- Postdoctoral training, The University of Chicago
Research Interests:
The Kinkel lab studies vertebrate developmental biology. Our focus is on the patterning of the endodermal germ layer, and we have a particular interest in development of the pancreas. Our work asks how pancreas cell types are specified, how organ size is determined, and how the pancreas is correctly localized within the embryo. We use the zebrafish model, and employ genetic tools including transgenic and mutant fish lines.
Much of our work centers around understanding development of the pancreatic beta cells. The beta cells are responsible for synthesizing and secreting insulin, and therefore have a major role in controlling blood sugar. Beta cell failure is the ultimate cause of all types of diabetes. Our motivation for these studies is that understanding how beta cells develop in the embryo will inform efforts to improve human health for diabetic patients.
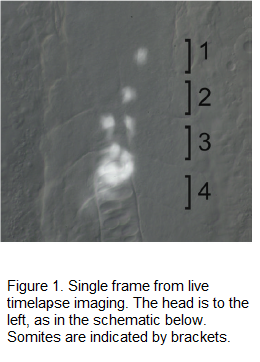
Figure 1 shows high magnification of the trunk at 19.5 hours post fertilization. Pancreatic beta-cells are labeled with Green Fluorescent Protein (GFP, here shown in white), using the Tg(ins:GFP) transgenic line. The live embryo was imaged using timelapse photography. The movie reveals that beta-cells differentiate in the anterior trunk, as single cells, then migrate posteriorly and cluster to form the pancreatic islet.
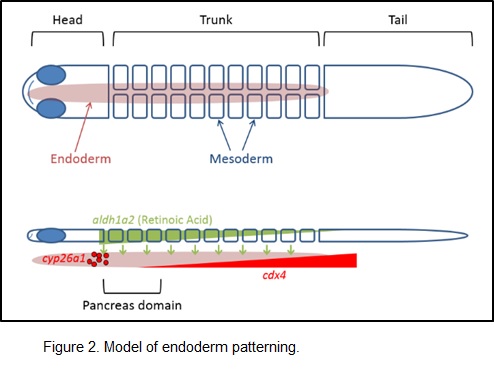
Our previous studies support the model of endoderm patterning shown in the schematic below. Briefly, retinoic acid (RA) is a positive signal that tells the endoderm to become pancreatic. RA is a small molecule that is synthesized in the overlying mesoderm, is secreted, and is received by nuclear receptors in the endoderm. Endodermal factors, including Cyp26 and Cdx4, limit the domain of endoderm that can become pancreatic in response to RA signaling. Therefore, Cyp26 and Cdx4 are critical for determining the size of the pancreas and its location. In ongoing work, we are focused on refining this model by integrating the role of the canonical Wnt signaling pathway.
Selected Publications:
Norton A, Franse KF, Daw, T, Gordon L, Vitiello PF, Kinkel MD. 2019. Larval rearing methods for small-scale production of healthy zebrafish. Eastern Biologist Special Issue 1:33-46.
Eames Nalle SC, Franse KF, Kinkel MD. 2017. Analysis of pancreatic disease in zebrafish. Methods in Cell Biology 138:271-295.
Eames SC, Kinkel MD, Rajan S, Prince VE, Philipson JH. 2013. Transgenic zebrafish model of the C43G human insulin mutation. J Diabetes Investig 4:157-167.
Eames SC, Philipson LH, Prince VE, and Kinkel MD. 2010. Blood sugar measurement in zebrafish reveals dynamics of glucose homeostasis. Zebrafish. 7:205-213.
Kinkel MD, Prince VE. 2009. On the Diabetic Menu: Zebrafish as a model for pancreas development and function. BioEssays. 31:139-152.
Kinkel MD, Sefton L, Prince VE. 2009. Cyp26 enzymes function in endoderm to regulate pancreatic field size. Proceedings of the National Academy of Sciences, USA., 106:7864-7869.
Kinkel MD, Eames SC, Alonzo MA, Prince VE. 2008. Cdx4 is required in the endoderm to localize the pancreas and limit beta-cell number. Development. 135:919-929.
Stafford D, White RJ, Kinkel MD, Linville A, Schilling TF, Prince VE. 2006. Retinoids signal directly to zebrafish endoderm to specify insulin-expressing beta-cells. Development. 133:949-956.
For a more complete list of publications and access to pdfs:

Title: Associate Professor, Developmental Biology
Department: Department of Biology
Email address: Email me
Phone: (828) 262-6907