Education and Professional Experience:
- BS, MS, PhD – Biology, Purdue University (Indianapolis, IN)
- Middle/High School Teaching Certification – Science, Biology, Chemistry (IN)
- Postdoctoral Fellow – Biochemistry, Dartmouth College (Hanover, NH)
- Research Scientist – Van Andel Institute (Grand Rapids, MI)
- Assistant Professor – Cancer Biology, Wake Forest University (Winston-Salem, NC)
Professional Organizations:
- American Association for Cancer Research
- American Society for Cell Biology
- The Invadosome Consortium
Teaching:
- Biological Concepts
- Cell Biology
- Cell Biology Laboratory
- Cancer Biology
Research:
My lab is interested in the cell signaling mechanisms that regulate cell motility, particularly in the context of tumor growth and metastasis. Our efforts are concentrated on the study of podosomes/invadopodia (i.e. invadosomes) – fine, actin-based cytoskeletal protrusions at the surface of professionally invasive cell types that enable adherence to and remodeling of the surrounding tissue. Our studies of the Src tyrosine kinase substrate and adaptor protein Tks5/Fish have been used to better define how podosomes and invadopodia form and function during the development and progression of cancer.
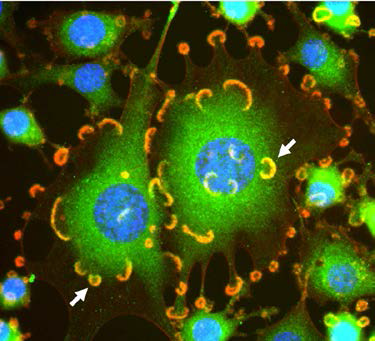
Src-transformed NIH3T3 cells showing co-localization (yellow/orange) of Tks5 (green) and F-actin (red) at invadosomes (arrows); nuclei (blue).
Podosomes/invadopodia are easily recognized by microscopy as punctate or rosette-shaped filamentous actin structures. They resemble the well-characterized focal contacts that enable cell-extracellular matrix adhesion, but are further distinguished by their ability to cluster proteases capable of extracellular matrix degradation. Podosomes are common to normal cell types with invasive properties including osteoclasts, macrophages, and vascular cells. Their discovery in cell lines derived from tumors, where they are referred to as invadopodia, suggests pathological functions as well. In fact, invadopodia were first discovered in fibroblasts transformed by the src oncogene, and Src signaling pathways appear to be a general contributor of invadopodia formation, and may also be the mechanism by which Src stimulates tumor growth and metastasis as well.
Tks5/Fish is an adaptor-type protein and a natural substrate of Src tyrosine kinase. Tks5 is associated with podosomes/invadopodia in Src-transformed fibroblasts, osteoclasts, macrophages, smooth muscle cells, endothelial cells, and in several cancer cell lines. This localization is driven by the enrichment of phosphoinositides at the membrane surface of these structures, which provide a target for the amino terminal, lipid-binding Phox (PX) homology domain of Tks5, as well as the lipid-binding domains of other podosome/invadopod marker proteins. Tks5 also has several other "handles" (SH3 domains, polyproline motifs, phosphorylated tyrosines) for protein-protein interactions, which likely enables Tks5-mediated scaffolding of podosome and invadopod machinery, including other adaptor (Grb, Nck), actin-polymerizing (WASp/N-WASp), and matrix-remodeling (ADAMs) proteins.
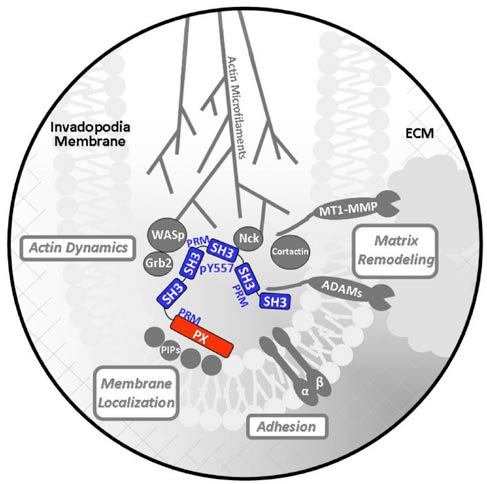
Cartoon of how the Src tyrosine kinase substrate and Tks5 adaptor protein (red & blue) may drive invadosome assembly and function
Our most recent work stretches across two fronts: (1) the role of invadopodia in cancer cell invasion, and (2) the role of podosomes in normal cell types of the tumor microenvironment that promote tumor progression. Tks5, for example, can induce invadopodia formation in prostate cancer cells. We also have recently published a paper on the role of Tks5 in macrophages, helping to establish the THP1 monocytic leukemia macrophage model system for future investigations of macrophage podosomes, and by extension, the role of Tks5 and tumor-associated macrophage podosomes in cancer cell invasion and metastasis.
Our guiding hypothesis is that Tks5 acts in a Src signaling pathway to scaffold a network of podosome-associated proteins, thereby engineering the necessary events of actin polymerization and proteolysis that contribute to podosome/invadopod formation and activity, cell invasion, and cancer development. Our future work will be to dissect the role of Tks5 in the formation and function of podosomes and invadopodia, in both normal and cancerous cells, in vitro and in vivo.
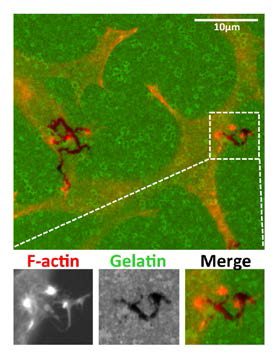
Tks5-overexpressing LNCaP prostate cancer cells produce invadopodia-like F-actin puncta (red) that co-localize with gelatin matrix (green) degradation (black).

Title: Associate Professor, Cancer Biology
Department: Department of Biology
Email address: Email me
Phone: (828) 262-2675